As biomolecules account for a growing share of drug discovery pathways in many pharmaceutical companies, there is growing interest in addressing development, production, and regulatory challenges. Since the potential immunogenicity of drugs is an important concern for both producers and regulators, how to define biopharmaceuticals is much more complex and potent than those of small molecule drugs. This, in turn, underscores the industry's urgent need for high-quality analytical tools that hopefully contribute to the full characterization of biopharmaceutical particles and agglomerates, as well as the characterization of the physical and chemical properties of the particles and contaminants in the drug.
What is the use of measurement?
The discovery of drug molecules from discovery to early formulation is a critical step. The physicochemical properties of the molecule are the determinants of drug formulation and administration; the earlier the physicochemical properties of the drug molecule are determined, the greater the economic benefit can be obtained – whether to determine the likelihood of success of the above steps, or to avoid possible early. failure. In this regard, it is desirable to be able to perform a series of non-destructive tests on very small samples and to focus more on improving the testing process that may be used.
From the physical and chemical characterization of drug molecules to the drug development process to the final product testing, the discovery and detection of protein agglomerates is the most important step in drug development because understanding the behavior of drug molecules in this aspect is the formulation, stability and safety of pharmaceutical products. It is all crucial. Protein structure is maintained by a combination of van der Waals forces, hydrogen bonds, disulfide bonds, and hydrophobic interactions. Changes in environmental conditions may affect some or all of these interaction forces - the result may cause abnormal folding of the agglomerates Or it has a negative impact on solubility. During this process, the activity of the protein often disappears, and it is possible that many agglomerates will develop immunogenicity, which will have a significant impact on the effectiveness and safety of the final therapeutic drug product.
Current regulatory expectations are for the characterization of agglomerates and insoluble particles ranging in size from "0.2-2" micron. Agglomerates larger than a few microns in size can be characterized visually. Less than this size, protein agglomerates can be characterized using sophisticated analytical techniques such as dynamic light scattering (DLS) and volume exclusion chromatography (SEC). Figure 1 shows the techniques and measurement ranges used in the above method. Resonance Mass Measurement (RMM) is the latest development of technology that is now also used to detect and count insoluble particles in the critical size range of 50 nm to 5 μm and to reliably support their buoyancy, net mass and particle size. Measurement. Resonance mass measurements provide a measurement window for insoluble particulates and submicron aggregates.
Use of dynamic light scattering
Dynamic light scattering is a fast, non-invasive measurement method that is particularly suitable for screening proteins in the early stages of drug development. This technique measures the intensity of scattered light produced by proteins under Brownian motion and converts the results into size and size distribution data. One of the greatest advantages of this technique is the ability to detect protein agglomerates at the earliest stages to determine the conditions under which the protein is formed or prevented from forming agglomerates.
The protein analysis process should select the ideal dynamic light scattering (DLS) technique. When the size of the protein in the solution is very small compared to the wavelength of the scattered light, the intensity of the scattered light is no longer dependent on the angle, which means that the same result can be obtained by measuring from any angle. However, as the protein size increases and exceeds ≈λ/10, the forward scattered light intensity begins to increase, and at this point, it becomes important to collect the data from a suitable angle. For example, using an instrument equipped with backscatter detection technology, the protein agglomeration process can be effectively monitored by measuring the scattered light intensity in both directions (see data below).
Volume exclusion chromatography
Volume Exclusion Chromatography (SEC), as the name suggests, separates dissolved polymers by the difference in molecular size of the fluid. This method is commonly used in biological science laboratories to characterize purified and recombinant proteins. When using SEC as an analytical tool, the amount and availability of information depends on the detection technique coupled to the chromatogram.
Single-detection SEC uses ultraviolet (UV) detectors to detect concentrations, which is still the traditional means of testing all protein agglomerates, but more powerful multi-detection systems are gaining popularity in this application because they can provide Complementary information and a comprehensive interpretation of the test results. For example, a system that combines a refractive index detector (RI), UV detector, light scattering detector, and viscosity detector to fully characterize protein samples, provide molecular weight data, and detailed structural changes and features while avoiding columns The need for calibration.
Figure 3 shows the detection of bovine serum albumin samples by the GPC /SEC chromatograph, which shows the response obtained by a system consisting of RI and light scattering detectors. The red color is the RI signal, and the reaction is more obvious at about 25 ml. The molecular weight calculated from the corresponding light scattering intensity data (green) can be determined to be a monomer. The two outflow peaks that appear earlier are determined to be dimers and trimers depending on the molecular weight. The large number of light scattering signals corresponding to the earliest peaks indicate that these are agglomerates. It is impossible to perform accurate qualitative and quantitative detection of all components if only relying on a single RI/UV detector system.
Resonance mass measurement
As a relatively new member of the analysis family, Resonance Mass Detection (RMM) detects and counts the amount of insoluble particles and submicron particles in a sample, while measuring its size and mass distribution. Since RMM is a counting technique, the above-mentioned mass distribution data is generated on a digital basis, thus giving the technique a good sensitivity to sparse particle groups.
At the heart of the resonance mass detector is a microelectromechanical system (MEMS) sensor with a resonant cantilever of micron-sized flow channels. When a single particle with a particle size range of 50 nm - 5 μm flows through the channel, the resonant frequency of the cantilever changes with the mass of the buoyancy of the particle, thereby inferring the dry mass and particle of the particle from the detected frequency change. path.
At the same time, MEMS sensors provide information on sample concentration, viscosity, density, and volume, as well as particles that detect and resolve positive and negative buoyancy relative to the solvent. The ability to distinguish between positive and negative buoyancy is useful, for example, he can simultaneously distinguish between silicone oil and protein components in samples contaminated with silicone oil droplets (see Figure 4). This is a problem that is often encountered in the later stages of drug development and in production, because silicone oil is a contaminant often encountered in syringes for drug delivery.
Another advantage of the resonance mass detection method is that it bridges the gap between visible and solution methods (such as volume exclusion chromatography, which is difficult to detect protein agglomeration). Regulators are keen to provide reliable quantification and comprehensive understanding of protein agglomerates. In order to meet the above analytical requirements, resonance mass spectrometry technology has emerged, which can penetrate the entire process of quality control, even in the presence of common pollutants. The situation can also meet the analytical needs.
Looking to the future
The inherent complexity and variability of biomolecules present new challenges to the rapidly evolving biopharmaceutical industry. It is important to use the prior art to detect protein particles or agglomerates in the finished product, but the industry is increasingly demanding various physical and chemical mechanisms that help to understand the above interactions and support the development of stable formulations. Measurement technology. Analytical solution providers must keep up with this pace of change and anticipate and respond to industry needs with the rapid development of instrumentation products that focus on measuring new and emerging “quality attributesâ€.
Reference material
1 Presentation: Susan Kirshner, FDA Office of Biotechnology. Download address: Click to download
Conclusion
Malvern Instruments, based in Pennsylvania, USA, recently launched a global bioscience development program. This is a resource-rich project focused on high-end technical collaborative development, working with many biopharmaceutical companies and prominent academic leaders in the field. The box below contains more details of the project. Please contact us if you need further information.
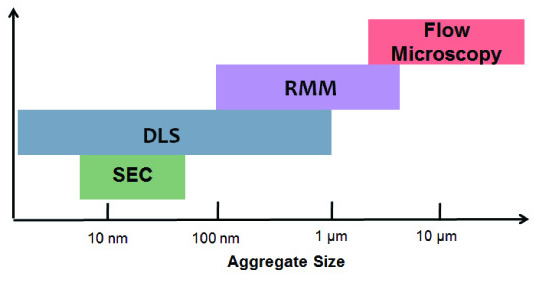
Figure 1: The industry needs a range of analytical techniques to ensure accurate measurement of all possible particle size ranges of protein aggregates
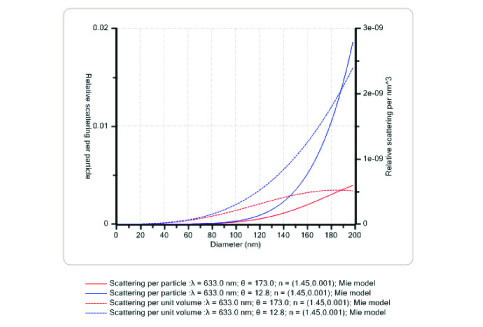
Figure 2: Simultaneous measurement of the light scattering intensity at the rear (173 degrees) and front (12.8 degrees) ensures that the particle size is accurately measured throughout the coacervation process.
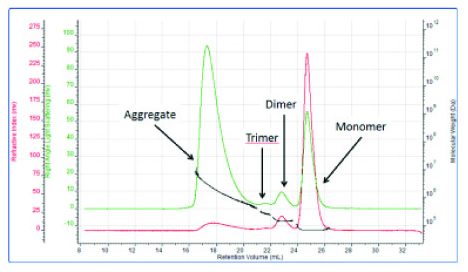
Figure 3: Measurement of bovine serum albumin using a GPC/SEC system consisting of RI and light scattering detectors.
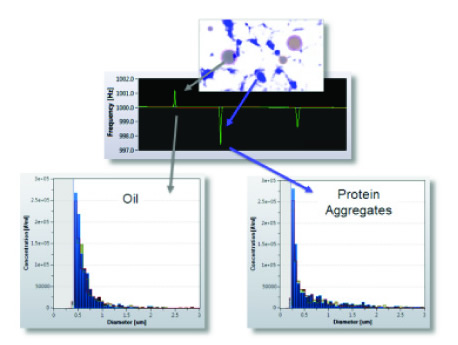
Figure 4: Resonance mass spectrometry (Archimedad, Malvern Instruments) performed two types of positive buoyant silicone oil droplets and negative buoyant protein agglomerated particles on a single protein sample.
The inch distribution is measured.
(Author: Stephen Ball ,, Malvern Instruments Product Marketing Manager)
Zhoushan Fudan Tourism CO., LTD , https://www.fudanfood.com